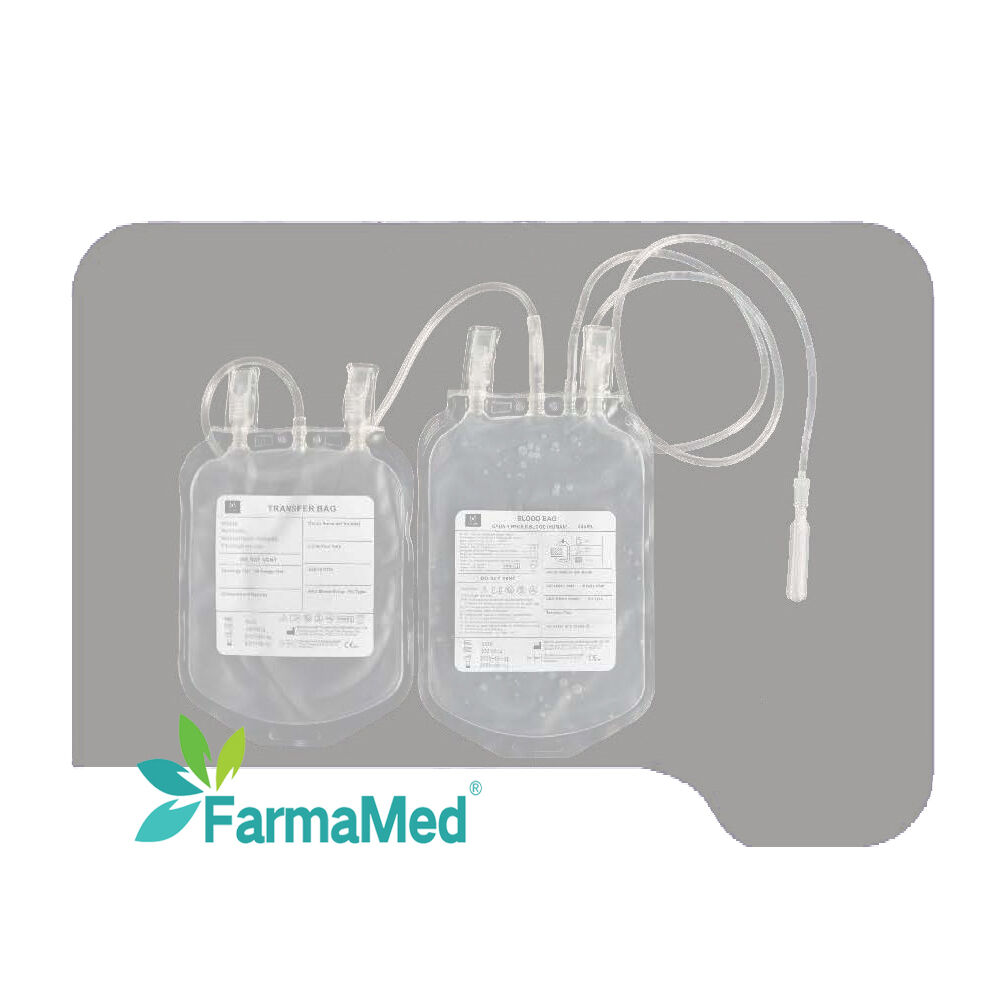
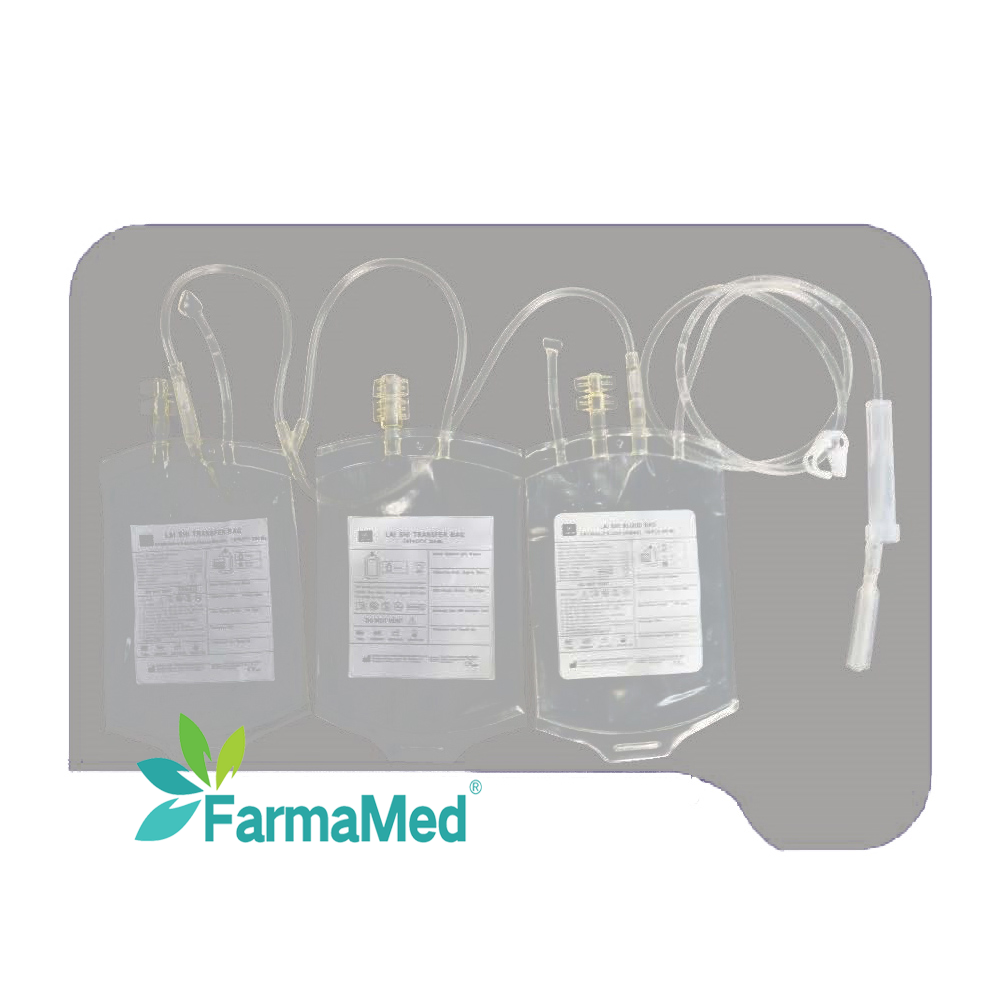
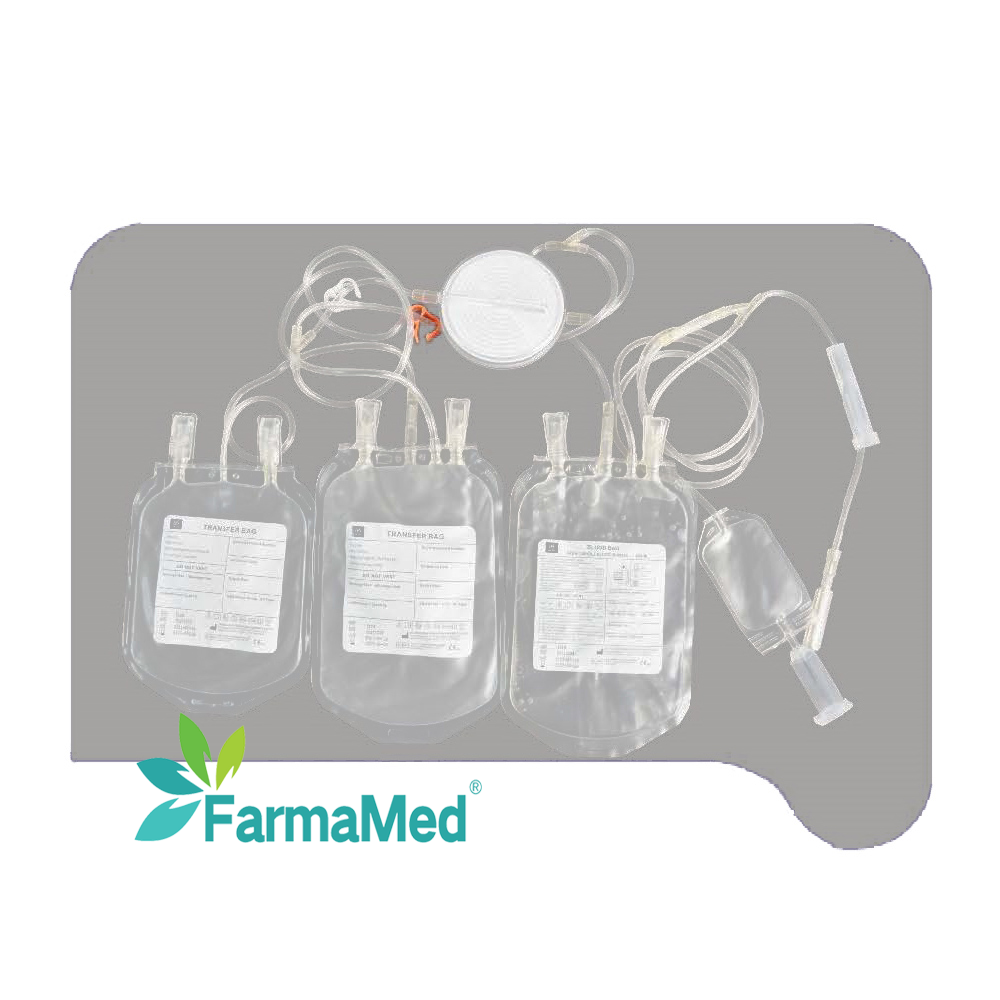
blood bag for hospital use
A blood bag for hospital use represents an essential medical device that serves as the cornerstone of modern transfusion medicine and blood storage systems. These specialized containers are meticulously engineered to collect, store, process, and transport blood and its components safely within healthcare facilities. The primary function of a blood bag for hospital use involves maintaining blood integrity while preventing contamination, coagulation, and bacterial growth during storage periods. These medical-grade containers utilize advanced polymer technology, typically constructed from polyvinyl chloride (PVC) or ethylene vinyl acetate (EVA) materials that ensure biocompatibility and chemical stability. The blood bag for hospital use incorporates multiple technological features including integrated anticoagulant solutions, sterile tubing systems, and secure closure mechanisms that maintain sterility throughout the collection and storage process. Modern blood bags feature multi-compartment designs that enable efficient separation of blood components such as red blood cells, plasma, and platelets. The technological sophistication of each blood bag for hospital use extends to specialized additives that extend storage life, preserve cellular function, and maintain optimal pH levels. Applications for blood bags span across emergency medicine, surgical procedures, cancer treatment, trauma care, and routine medical interventions requiring blood transfusions. Healthcare professionals rely on these devices for autologous blood collection, where patients donate their own blood prior to surgery, and for managing blood inventories in hospital blood banks. The blood bag for hospital use also supports specialized procedures like plasmapheresis, where plasma is separated and collected for therapeutic purposes. Quality control measures ensure each blood bag for hospital use meets stringent regulatory standards established by health authorities worldwide, guaranteeing patient safety and product reliability in critical medical situations.