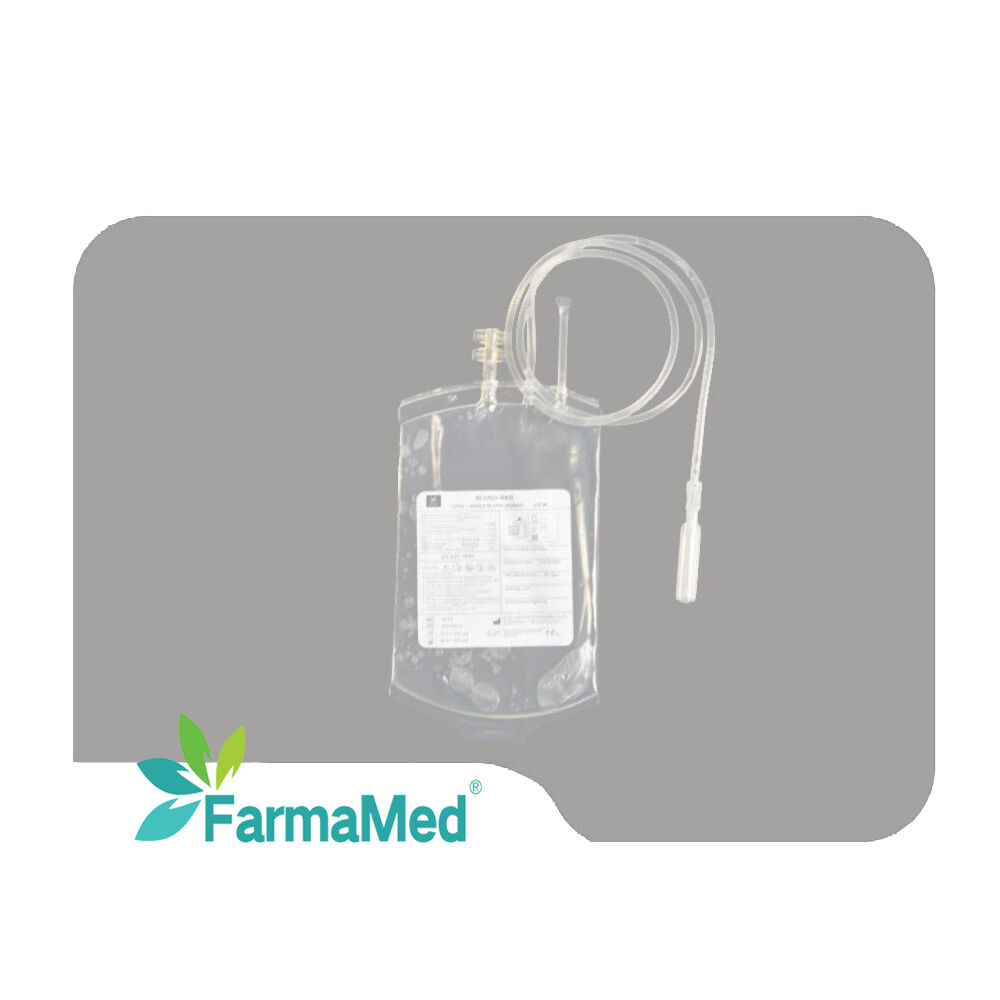
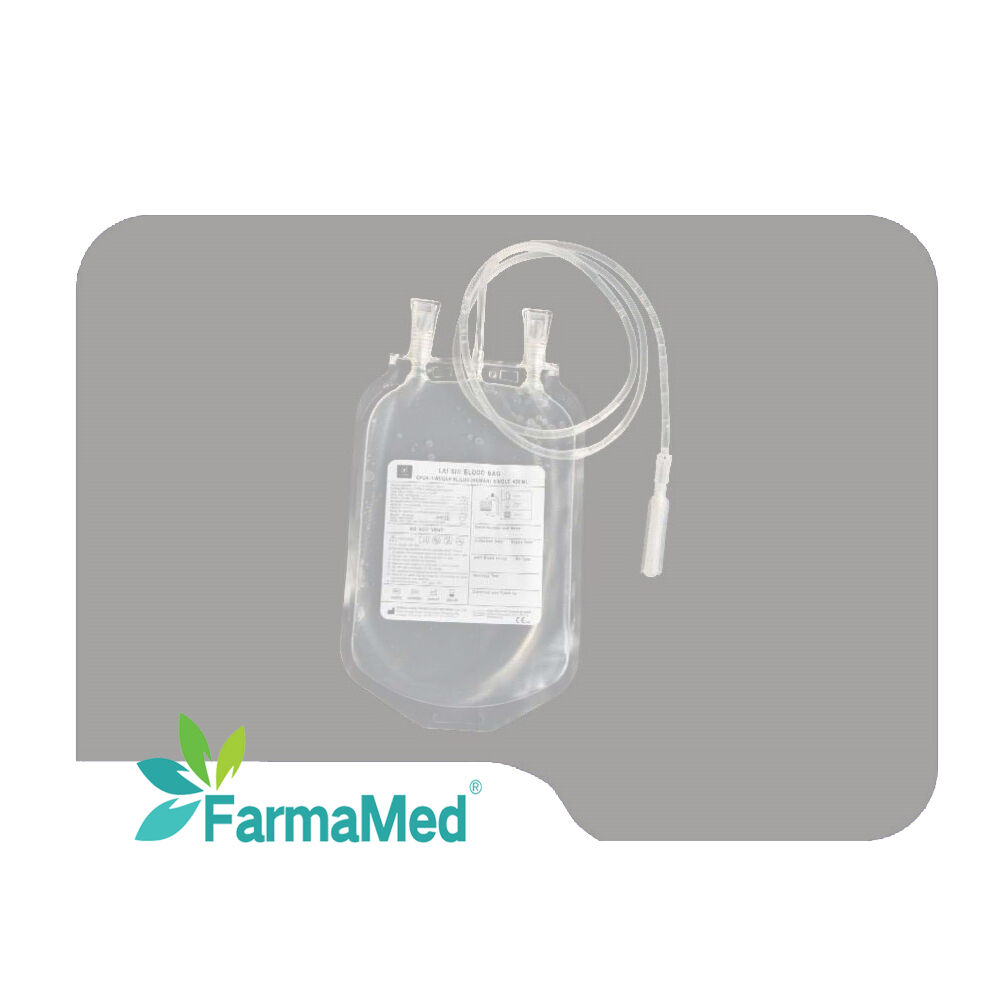
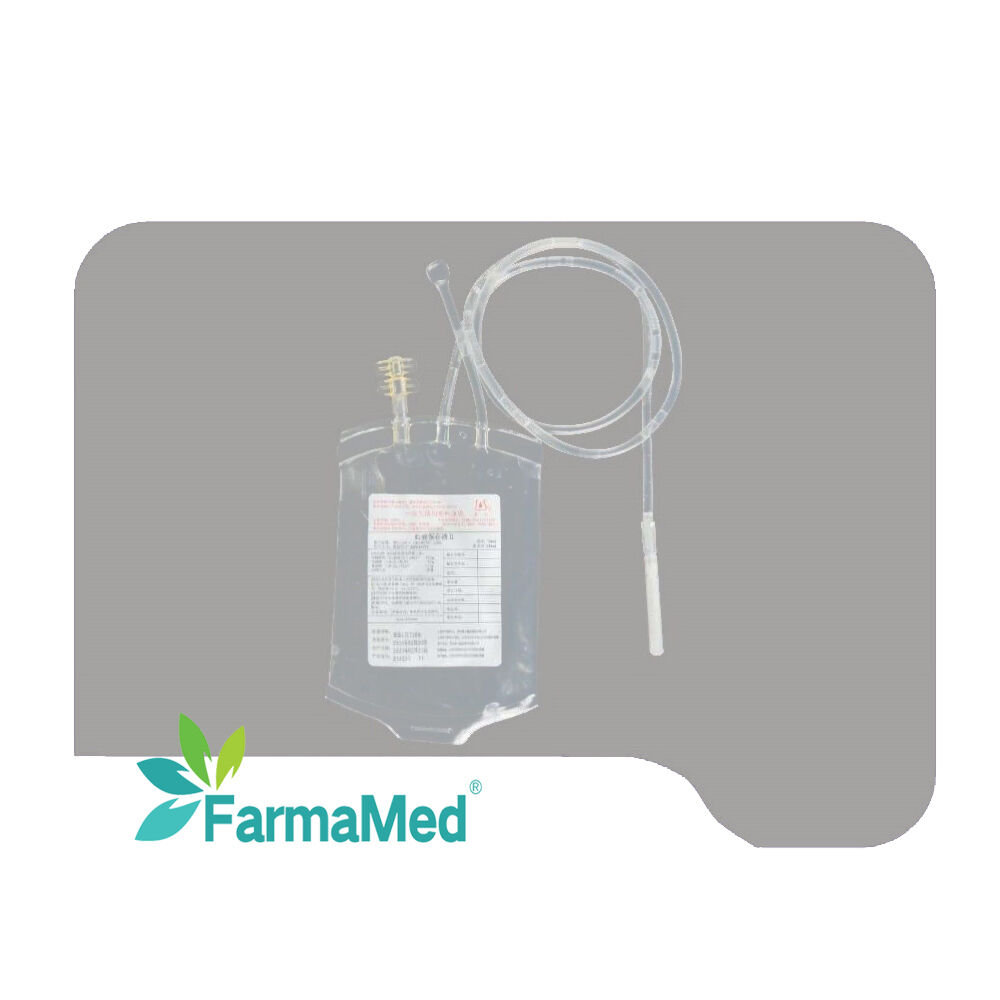
blood bag factory
A blood bag factory represents a critical component of the global healthcare infrastructure, serving as a specialized manufacturing facility dedicated to producing sterile, safe, and reliable blood collection and storage systems. These sophisticated production centers utilize advanced medical-grade materials and cutting-edge manufacturing processes to create blood bags that meet stringent international quality standards and regulatory requirements. The primary function of a blood bag factory involves the systematic production of various types of blood collection containers, including single, double, triple, and quadruple bag systems designed for different blood processing needs. Modern blood bag factory operations incorporate state-of-the-art clean room environments, automated production lines, and comprehensive quality control systems to ensure product integrity and patient safety. The technological features of a contemporary blood bag factory include precision welding equipment for creating leak-proof seals, automated testing systems for bag strength and sterility verification, and sophisticated packaging machinery that maintains product sterility throughout the supply chain. These facilities employ advanced polymer science to develop blood bag materials that prevent hemolysis, maintain blood viability, and provide optimal storage conditions for extended periods. Applications for blood bag factory products span across multiple healthcare sectors, including hospitals, blood banks, research laboratories, and emergency medical services. The versatility of modern blood bag systems enables efficient whole blood collection, component separation, plasma processing, and long-term storage solutions. Quality assurance protocols within a blood bag factory encompass rigorous testing procedures, including biocompatibility assessments, sterility validation, and performance evaluations under various environmental conditions. The integration of digital monitoring systems and automated quality control measures ensures consistent product excellence while minimizing human error and contamination risks. Environmental considerations have become increasingly important in blood bag factory operations, with many facilities implementing sustainable manufacturing practices, waste reduction programs, and eco-friendly packaging solutions that align with global environmental protection initiatives.