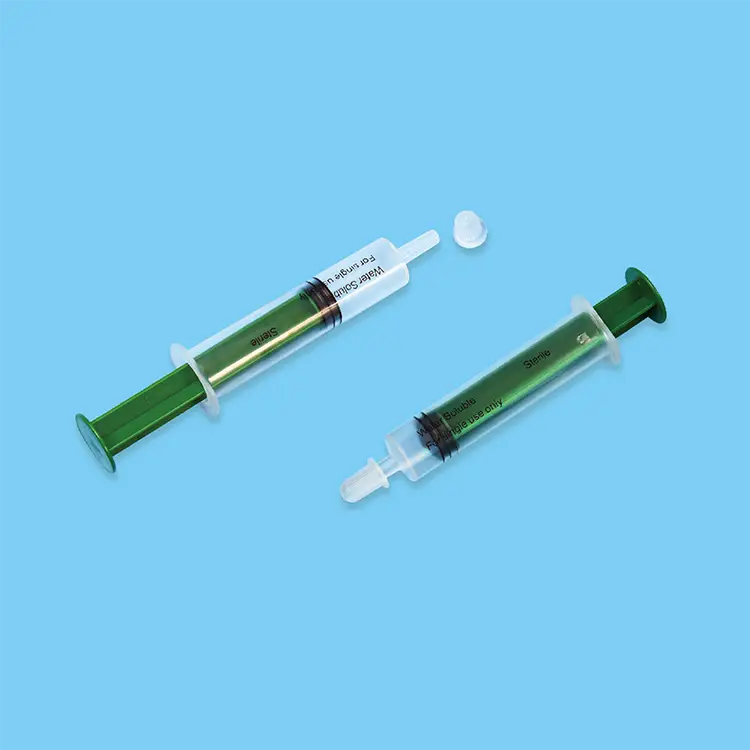
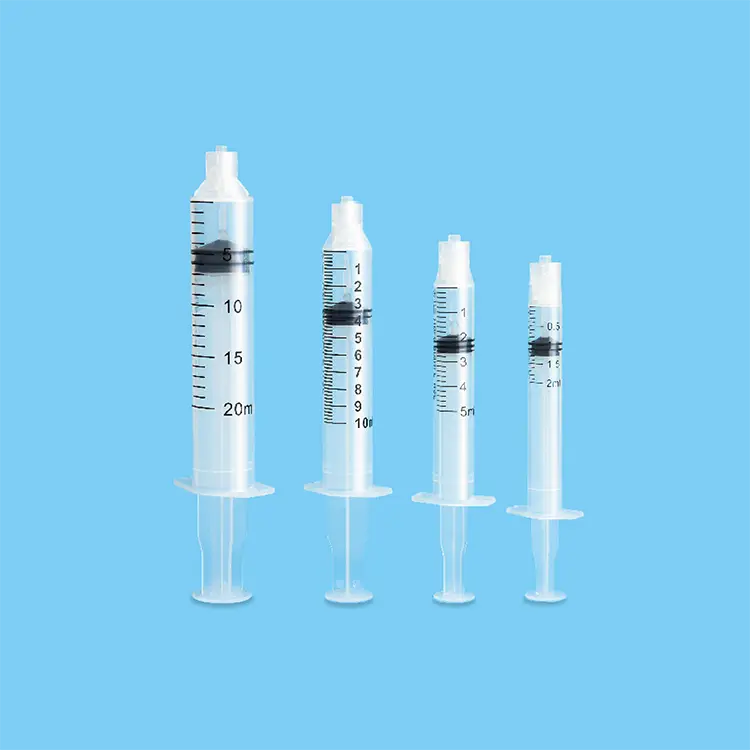
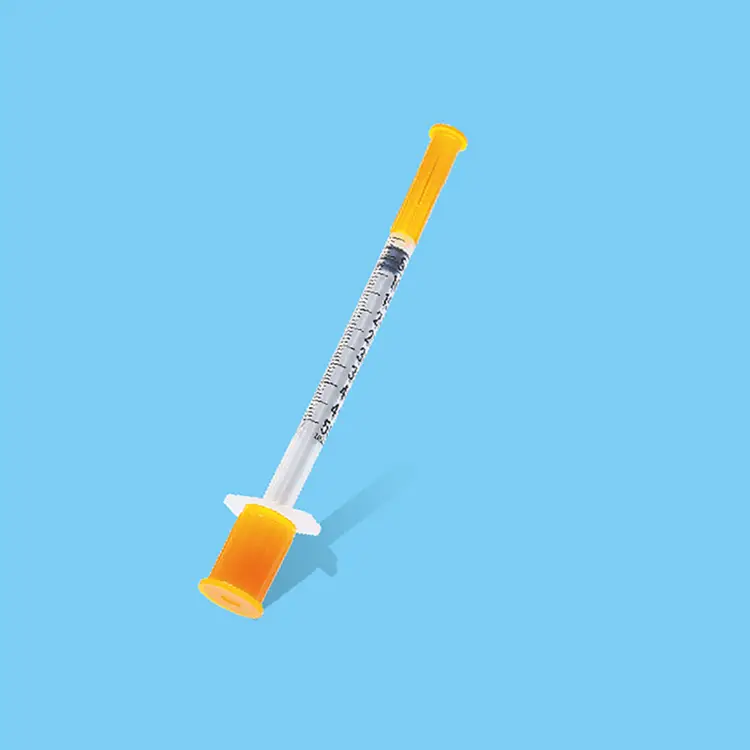
sterile needles and syringes
Sterile needles and syringes represent essential medical devices engineered to deliver safe, contamination-free healthcare solutions across diverse clinical environments. These precision-manufactured instruments undergo rigorous sterilization processes using advanced gamma radiation, ethylene oxide, or steam sterilization methods to eliminate all microorganisms, bacteria, viruses, and pathogens. The primary functions of sterile needles and syringes encompass medication administration, vaccine delivery, blood sampling, insulin injection, and various therapeutic procedures requiring sterile access to the human body. Modern sterile needles and syringes incorporate cutting-edge technological features including ultra-sharp beveled needle tips that minimize tissue trauma and patient discomfort during penetration. The syringe barrels utilize medical-grade polypropylene or polystyrene materials that ensure chemical compatibility with pharmaceutical compounds while maintaining structural integrity under pressure. Advanced lubrication coatings on needle surfaces reduce insertion force and enhance patient comfort during procedures. Safety mechanisms such as retractable needles, safety shields, and locking systems prevent accidental needlestick injuries and cross-contamination risks. Applications for sterile needles and syringes span multiple healthcare sectors including hospitals, clinics, laboratories, emergency medical services, home healthcare, veterinary practices, and pharmaceutical research facilities. Healthcare professionals rely on these devices for routine immunizations, chronic disease management, emergency interventions, diagnostic procedures, and specialized treatments. The manufacturing process involves automated assembly lines with stringent quality control protocols, ensuring consistent performance and reliability. Each sterile needle and syringe package includes detailed specifications regarding gauge sizes, volume capacities, and expiration dates to guarantee optimal clinical outcomes and patient safety standards throughout various medical applications.