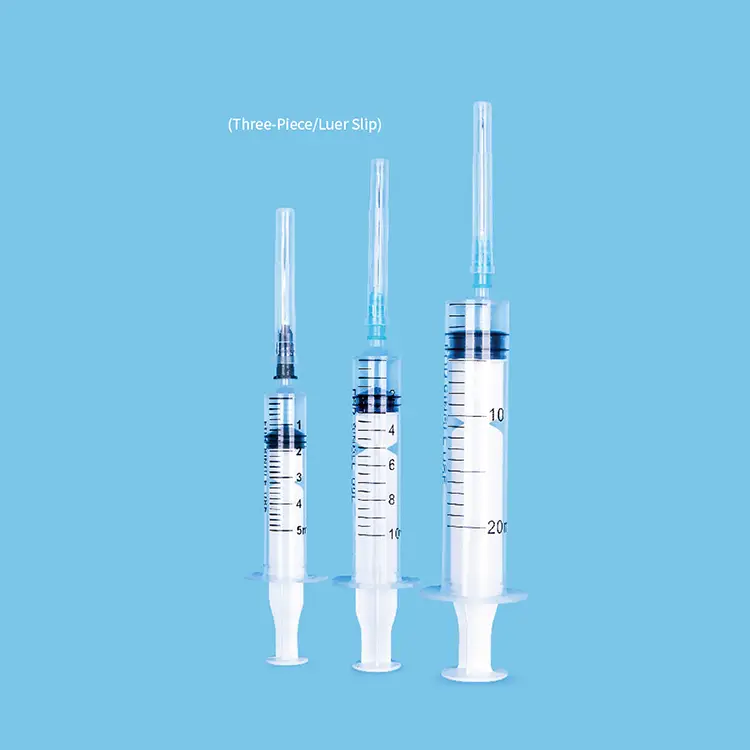
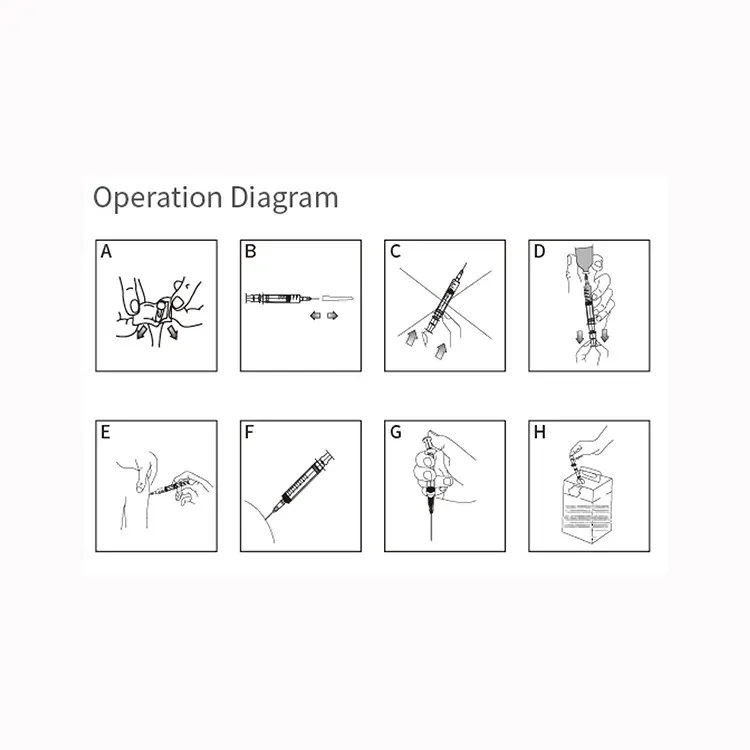
syringes and needles factory
A syringes and needles factory represents a sophisticated medical device manufacturing facility that specializes in producing sterile injection equipment essential for healthcare delivery worldwide. These specialized manufacturing plants combine advanced automation technology with stringent quality control systems to create precise medical instruments that meet international safety standards. The primary function of a syringes and needles factory involves the mass production of disposable syringes ranging from insulin syringes to large-volume injection devices, alongside various needle types including hypodermic needles, safety needles, and specialized surgical needles. Modern syringes and needles factory operations utilize state-of-the-art injection molding machines that shape medical-grade plastics into syringe barrels, plungers, and needle hubs with exceptional precision. The technological backbone of any syringes and needles factory includes automated assembly lines equipped with robotic systems that ensure consistent product quality while maintaining sterile conditions throughout the manufacturing process. These facilities employ sophisticated sterilization methods such as ethylene oxide gas sterilization or gamma radiation to eliminate potential contaminants and guarantee product safety. Quality assurance laboratories within each syringes and needles factory conduct rigorous testing protocols including dimensional accuracy checks, sterility testing, and biocompatibility assessments to ensure compliance with FDA, CE, and ISO standards. The applications of products manufactured in a syringes and needles factory span across multiple healthcare sectors including hospitals, clinics, vaccination centers, veterinary practices, and home healthcare settings. Each syringes and needles factory typically produces millions of units annually, supporting global healthcare initiatives and emergency response programs while maintaining cost-effective production schedules that benefit both manufacturers and end-users seeking reliable medical injection equipment.