syringes and needles exporter
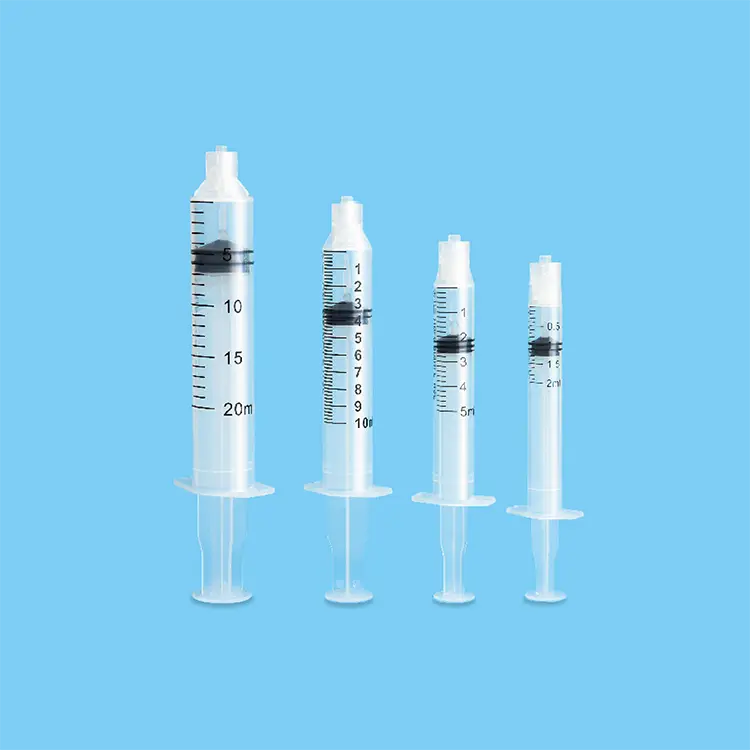
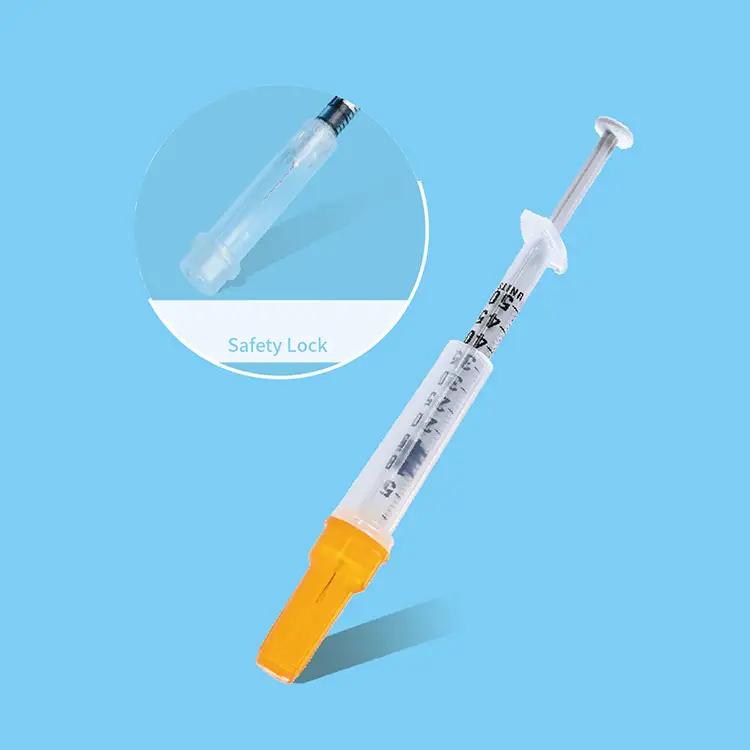
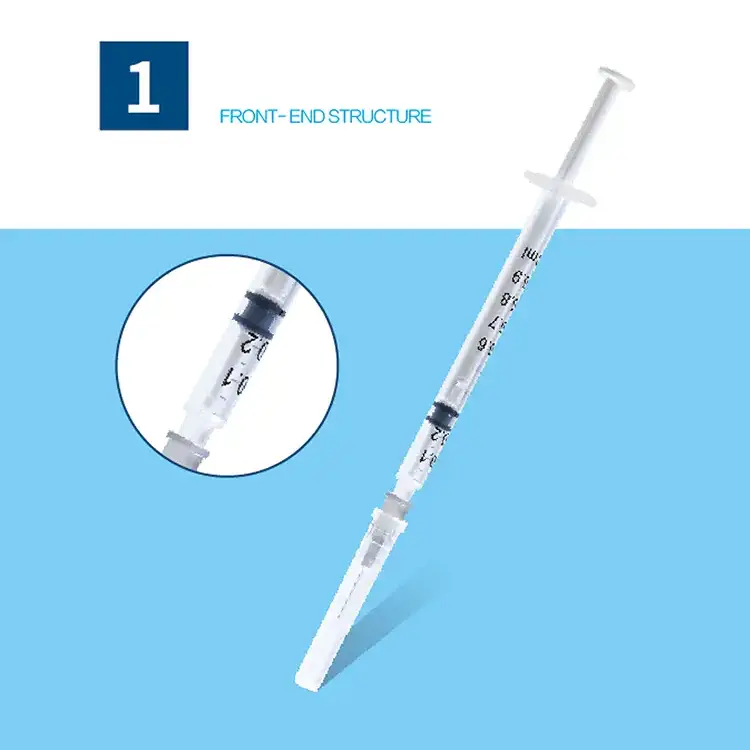
A syringes and needles exporter plays a crucial role in the global healthcare supply chain, serving as a vital bridge between manufacturers and healthcare providers worldwide. These specialized companies focus on sourcing, quality assurance, and international distribution of medical injection devices that meet stringent regulatory standards across different markets. The primary function of a syringes and needles exporter involves establishing robust procurement networks with certified manufacturers, ensuring compliance with international medical device regulations, and maintaining cold chain logistics for temperature-sensitive products. Modern exporters utilize advanced inventory management systems that track product batches, expiration dates, and regulatory certifications in real-time. Their technological infrastructure includes automated quality control processes, digital documentation systems for regulatory compliance, and sophisticated logistics platforms that optimize shipping routes and delivery schedules. The applications of syringes and needles exporter services span across hospitals, clinics, vaccination centers, research laboratories, veterinary practices, and pharmaceutical companies requiring reliable access to sterile injection equipment. These exporters specialize in various product categories including disposable syringes, safety syringes, insulin syringes, tuberculin syringes, hypodermic needles, safety needles, and specialized injection devices for specific medical procedures. Their operations encompass regulatory affairs management, where teams ensure all exported products meet destination country requirements, including FDA approvals, CE markings, and ISO certifications. The technological features of leading exporters include blockchain-based traceability systems that provide complete product history from manufacture to delivery, automated reordering systems that prevent stockouts, and integrated quality management systems that monitor product integrity throughout the supply chain. Additionally, these companies maintain comprehensive databases of international regulatory requirements, enabling seamless market entry for their products while ensuring healthcare providers receive consistent, high-quality medical devices that support safe patient care practices.