disposable syringes and needles supplier
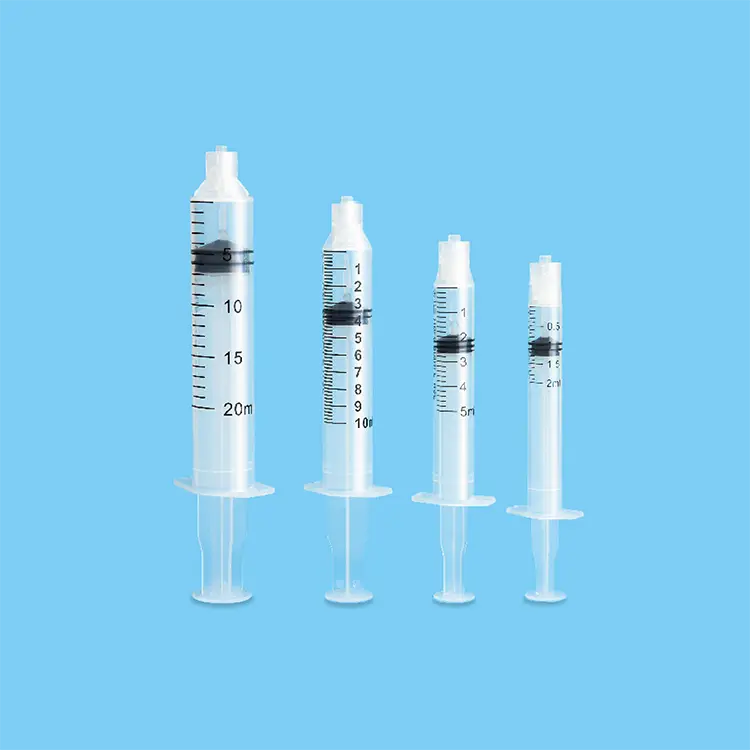
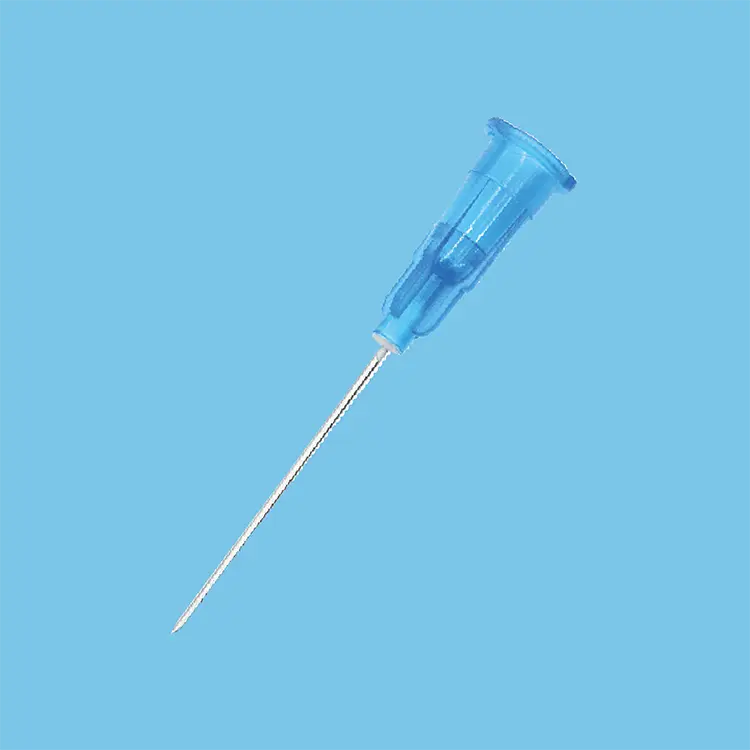
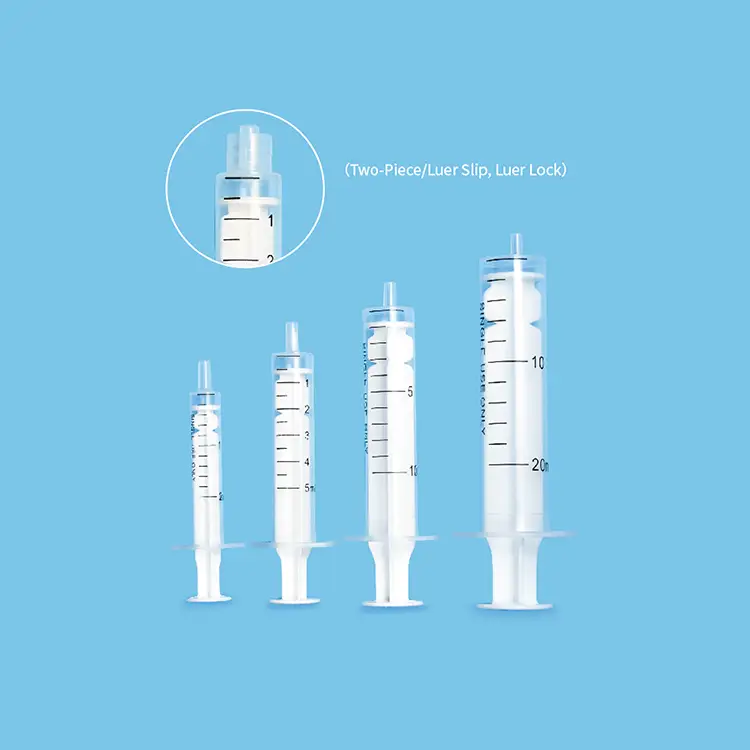
A disposable syringes and needles supplier represents a crucial healthcare partner that specializes in manufacturing and distributing single-use injection equipment designed for medical, pharmaceutical, and research applications. These suppliers focus on producing sterile, precision-engineered devices that ensure patient safety while meeting stringent regulatory standards across global healthcare markets. The primary function of a disposable syringes and needles supplier centers on delivering consistent, reliable injection solutions that eliminate cross-contamination risks inherent in reusable alternatives. Modern suppliers integrate advanced manufacturing technologies including automated assembly lines, laser-etched graduations, and multi-layer quality control systems to guarantee product integrity. Technological features encompass silicone-coated plungers for smooth operation, tri-beveled needle points for reduced patient discomfort, and tamper-evident packaging that maintains sterility until point of use. These suppliers utilize medical-grade polypropylene and stainless steel materials that comply with international standards such as ISO 13485 and FDA regulations. Applications span across vaccination programs, blood collection procedures, insulin administration, emergency medical services, veterinary practices, and laboratory testing environments. The disposable syringes and needles supplier ecosystem includes various syringe capacities ranging from 0.5ml to 60ml configurations, accommodating diverse clinical requirements from pediatric vaccinations to large-volume injections. Safety-engineered products featuring retractable needles and auto-disable mechanisms represent innovative technological advancements that prevent needlestick injuries and unauthorized reuse. Supply chain management involves temperature-controlled storage facilities, lot tracking systems, and distribution networks that ensure global accessibility while maintaining product efficacy. These suppliers also provide customization options including specific needle gauges, specialized tip designs, and private labeling services to meet unique customer specifications and regulatory requirements across different geographical markets.