hypodermic syringes and needles
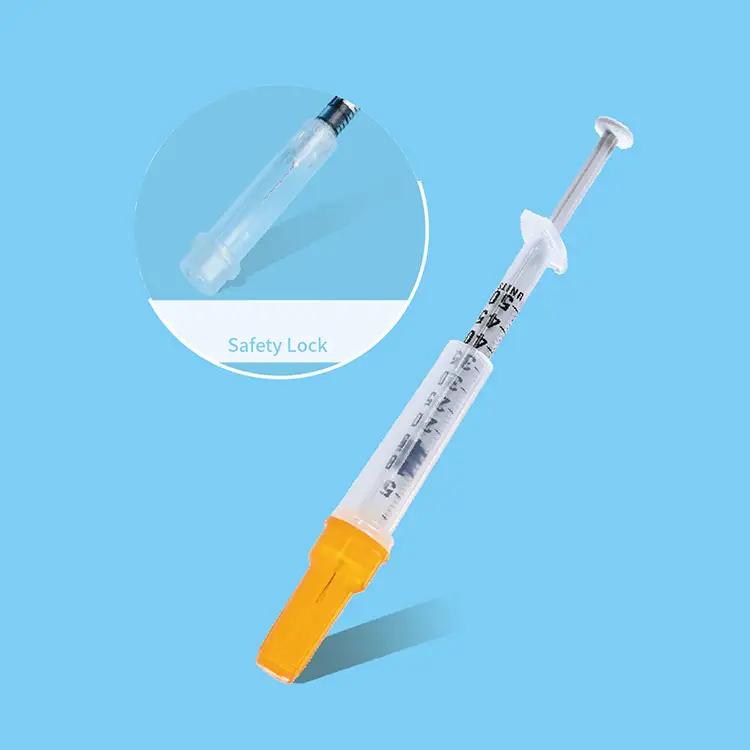
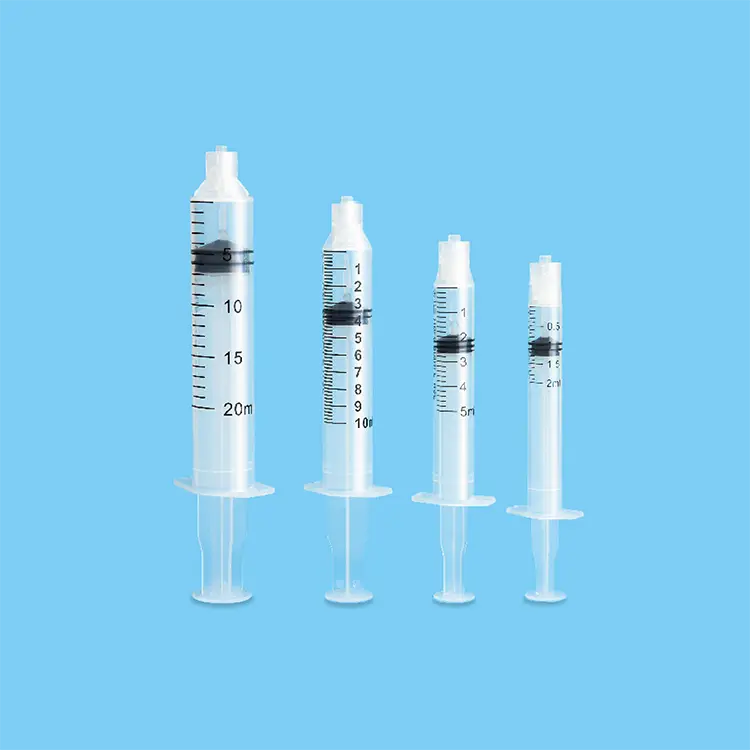
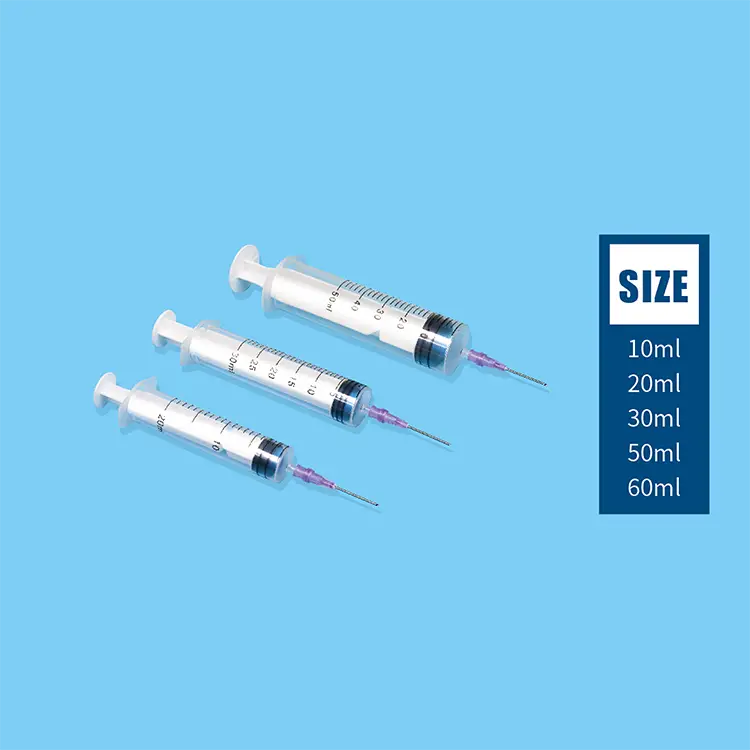
Hypodermic syringes and needles represent fundamental medical instruments designed for precise fluid injection and extraction procedures across healthcare environments. These specialized devices consist of a graduated barrel, plunger mechanism, and sterile needle attachment that enables healthcare professionals to deliver medications, vaccines, and therapeutic substances directly into patient tissues. The primary functions of hypodermic syringes and needles encompass subcutaneous injections, intramuscular administrations, intravenous deliveries, and specimen collection procedures. Modern hypodermic syringes and needles incorporate advanced engineering technologies including ultra-sharp needle tips manufactured through precision grinding processes, ensuring minimal tissue trauma during penetration. The barrel construction utilizes medical-grade polypropylene materials that provide chemical resistance and dimensional stability under varying temperature conditions. Technological features of contemporary hypodermic syringes and needles include safety mechanisms such as retractable needle systems, needle guards, and engineered sharps injury protection devices that significantly reduce accidental needlestick incidents. Applications for hypodermic syringes and needles span diverse medical specialties including emergency medicine, pediatric care, geriatric treatment, surgical procedures, diagnostic testing, and preventive immunization programs. Hospital settings rely extensively on hypodermic syringes and needles for patient care protocols, while outpatient clinics utilize these instruments for routine medical procedures and chronic disease management. Veterinary practices also depend on specialized hypodermic syringes and needles designed for animal treatment applications. The manufacturing standards for hypodermic syringes and needles adhere to stringent regulatory requirements established by health authorities, ensuring consistent product quality, sterility maintenance, and patient safety compliance. These medical devices undergo rigorous testing procedures including biocompatibility assessments, sterility validation, and performance verification protocols before market distribution.