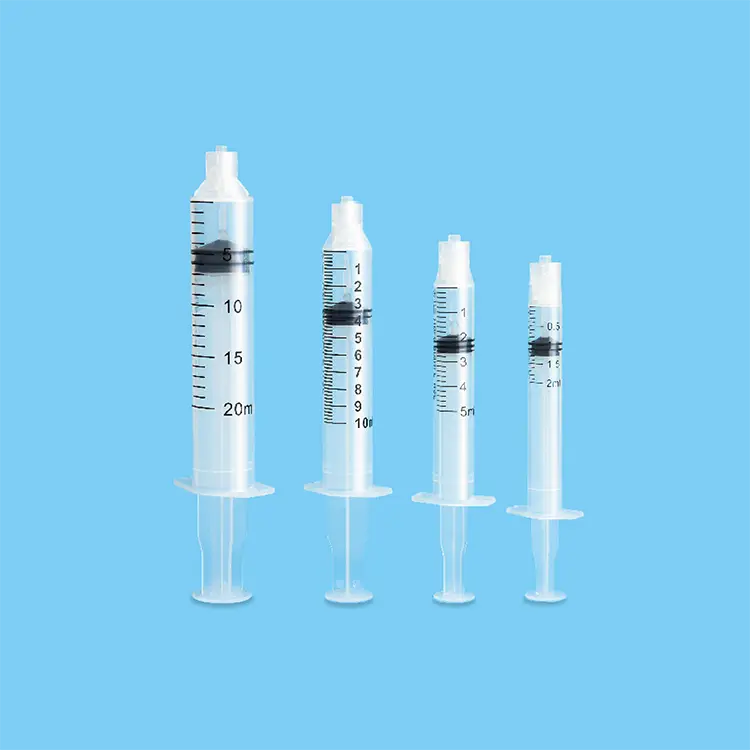
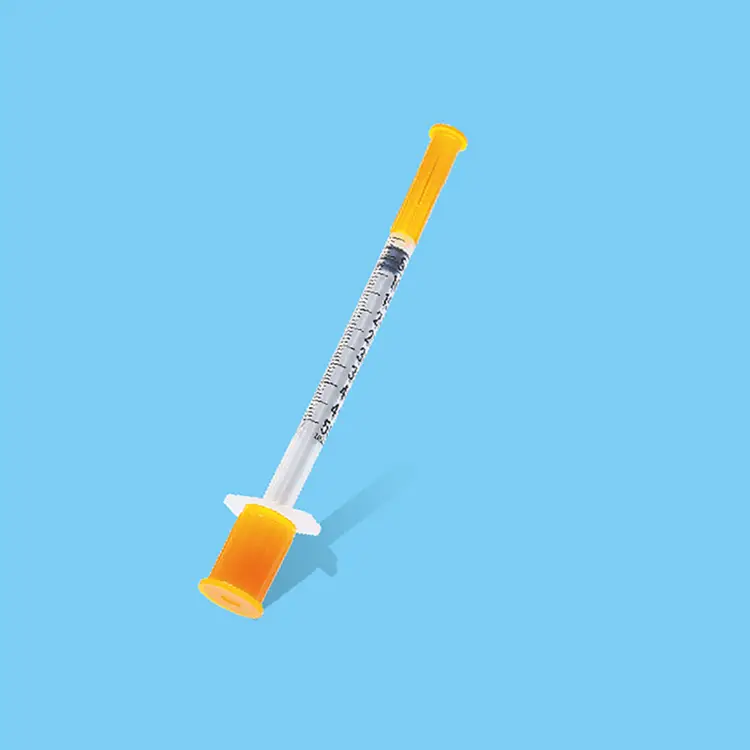
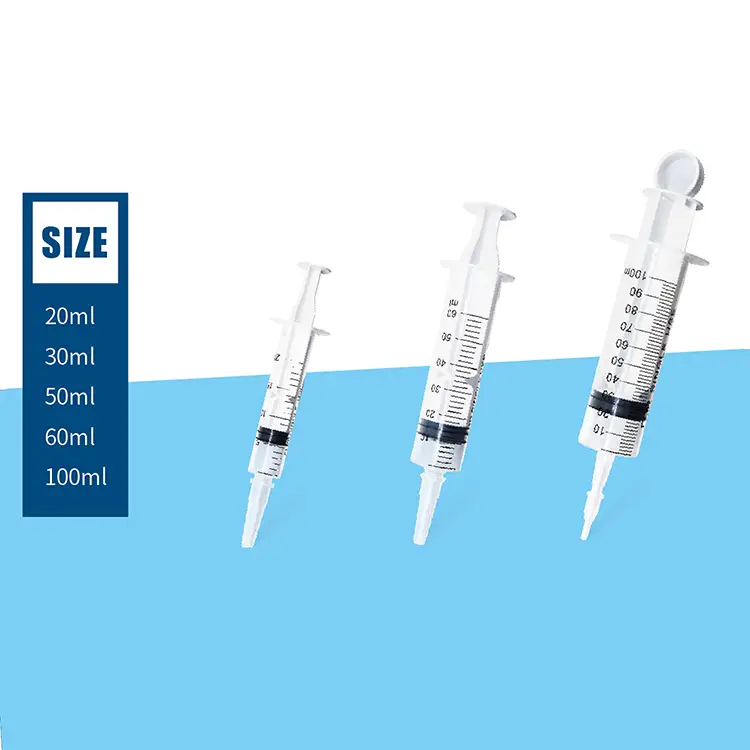
syringes and needles oem
Syringes and needles OEM manufacturing represents a critical component of the global healthcare supply chain, providing customized medical devices that meet specific requirements for healthcare providers, pharmaceutical companies, and medical device distributors worldwide. OEM services in this sector focus on producing high-quality injection devices that comply with international standards while offering flexibility in design, materials, and specifications. The primary functions of syringes and needles OEM operations encompass complete product development cycles, from initial concept design through final packaging and distribution. These services include precision manufacturing of various syringe types, including disposable syringes, safety syringes, prefilled syringes, and specialty injection devices. The needle component manufacturing covers different gauge sizes, lengths, and specialized configurations for specific medical applications. Technological features of modern syringes and needles OEM facilities incorporate advanced automation systems, cleanroom environments, and rigorous quality control processes that ensure consistent product performance and safety. State-of-the-art injection molding equipment produces syringe barrels and plungers with precise tolerances, while sophisticated needle manufacturing processes create sharp, consistent penetration points that minimize patient discomfort. Applications for syringes and needles OEM products span across multiple healthcare sectors, including hospitals, clinics, vaccination programs, diabetes management, emergency medical services, and home healthcare settings. The versatility of OEM manufacturing allows for customization based on specific regional requirements, regulatory compliance needs, and unique application demands. Quality assurance protocols integrated into syringes and needles OEM operations ensure that every product meets stringent medical device regulations, including FDA approvals, CE marking, and ISO certifications. These comprehensive manufacturing solutions provide healthcare organizations with reliable, cost-effective injection devices that maintain the highest standards of safety and performance while supporting various medical procedures and patient care initiatives across diverse healthcare environments.