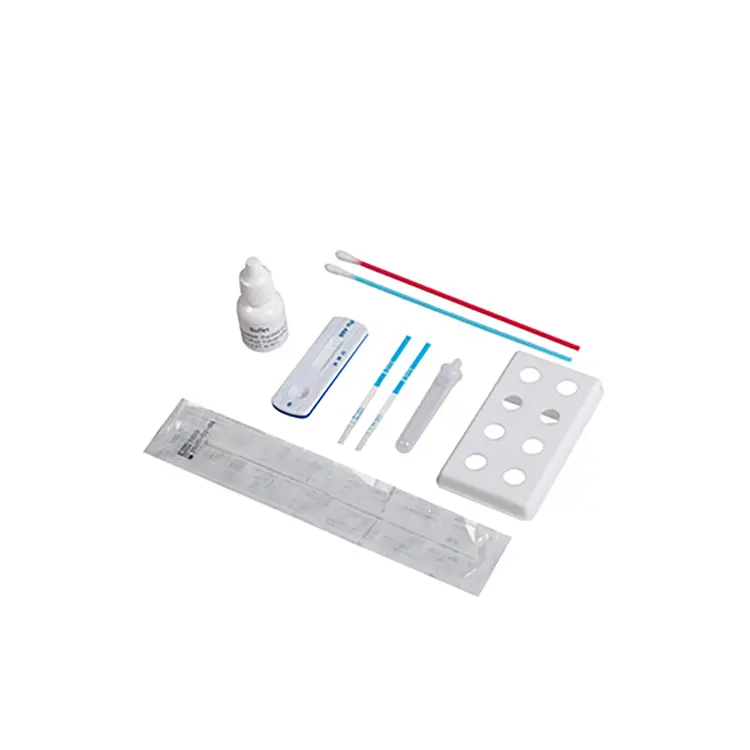
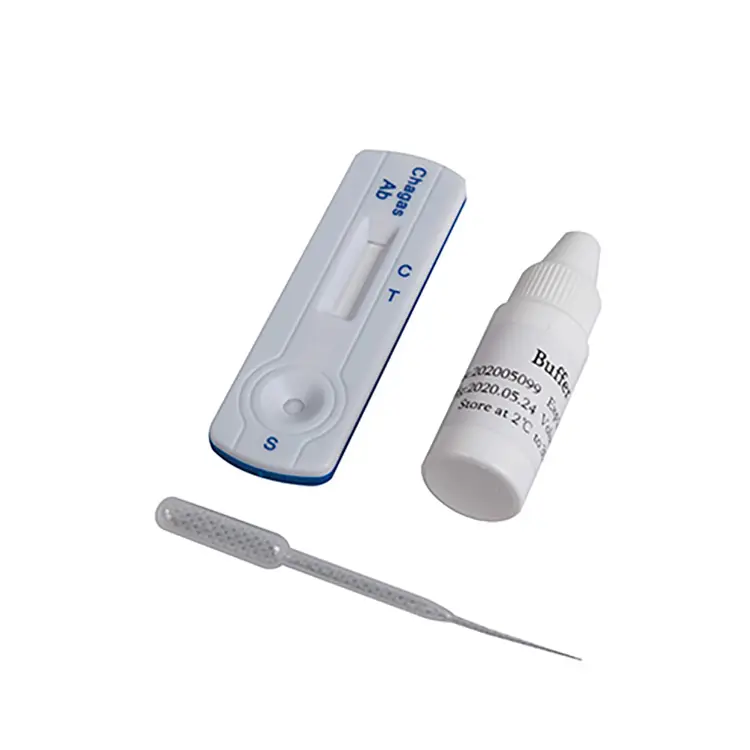
rapid diagnostic test manufacturer
A rapid diagnostic test manufacturer stands at the forefront of modern healthcare innovation, delivering cutting-edge solutions that transform patient care through immediate, accurate testing capabilities. These specialized companies develop, produce, and distribute diagnostic devices that provide healthcare professionals with critical medical information within minutes rather than days. The rapid diagnostic test manufacturer operates sophisticated facilities equipped with advanced biotechnology, precision engineering systems, and stringent quality control measures to ensure every product meets international medical standards. Their primary function revolves around creating point-of-care testing solutions that enable healthcare providers to make informed decisions quickly, ultimately improving patient outcomes and reducing healthcare costs. The technological infrastructure employed by a rapid diagnostic test manufacturer incorporates state-of-the-art immunoassay techniques, molecular diagnostics, and lateral flow technology. These systems utilize antibody-antigen interactions, enzymatic reactions, and fluorescent markers to detect specific biomarkers, pathogens, or medical conditions with remarkable precision. Manufacturing processes involve cleanroom environments, automated production lines, and rigorous validation protocols that guarantee consistent product quality and reliability. Applications for products from a rapid diagnostic test manufacturer span numerous medical fields including infectious disease detection, cardiac markers, pregnancy testing, drug screening, and chronic disease monitoring. Healthcare settings such as hospitals, clinics, emergency departments, laboratories, and remote medical facilities rely heavily on these rapid testing solutions. The versatility of these diagnostic tools makes them invaluable in diverse scenarios from routine health screenings to emergency medical situations where time-sensitive decisions can save lives. Each rapid diagnostic test manufacturer focuses on developing user-friendly devices that require minimal training while delivering laboratory-quality results at the point of care.