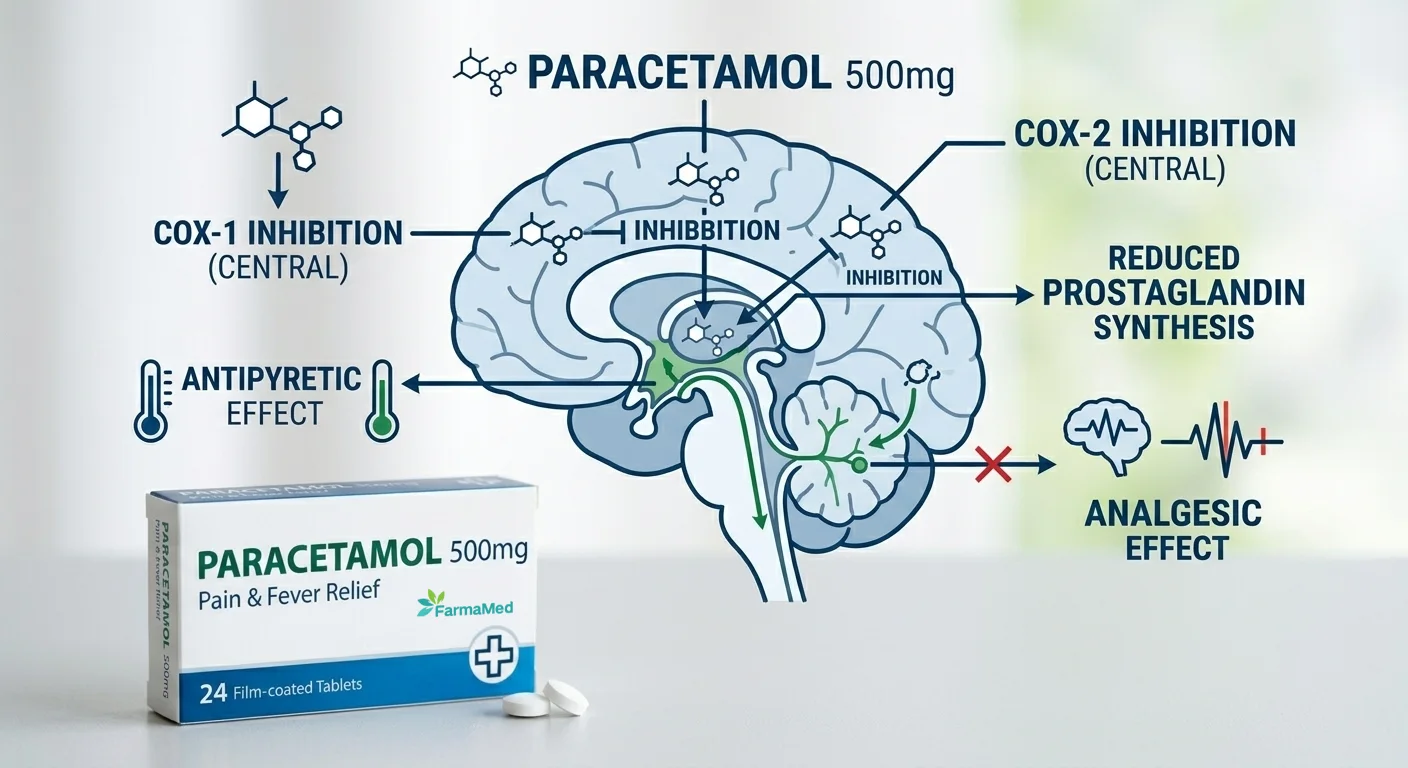
paracetamol tablets manufacturer
A paracetamol tablets manufacturer operates as a specialized pharmaceutical company dedicated to producing acetaminophen-based medications that serve millions of patients worldwide. These manufacturers combine advanced pharmaceutical technology with stringent quality control systems to create safe, effective pain relief and fever reduction solutions. The primary function of a paracetamol tablets manufacturer involves the precise formulation, production, and packaging of acetaminophen in tablet form, ensuring consistent dosage and therapeutic efficacy. Modern paracetamol tablets manufacturers utilize state-of-the-art equipment including high-speed tablet presses, coating machines, and automated packaging systems that maintain product integrity throughout the manufacturing process. The technological features employed by leading paracetamol tablets manufacturers include continuous monitoring systems, real-time quality assessment tools, and computer-controlled production lines that guarantee batch consistency and regulatory compliance. These manufacturers implement comprehensive testing protocols covering raw material verification, in-process monitoring, and finished product analysis to meet international pharmaceutical standards. Applications for paracetamol tablets span across healthcare sectors, from hospital pharmacies to retail drugstores, serving patients requiring pain management and fever control. The paracetamol tablets manufacturer must maintain strict adherence to Good Manufacturing Practices (GMP) while ensuring scalable production capabilities to meet market demand fluctuations. Advanced manufacturers incorporate environmental sustainability measures, waste reduction programs, and energy-efficient processes into their operations. The integration of digital technologies enables paracetamol tablets manufacturers to optimize production schedules, track inventory levels, and maintain complete traceability from raw materials to finished products, ensuring patient safety and regulatory compliance across all distribution channels.