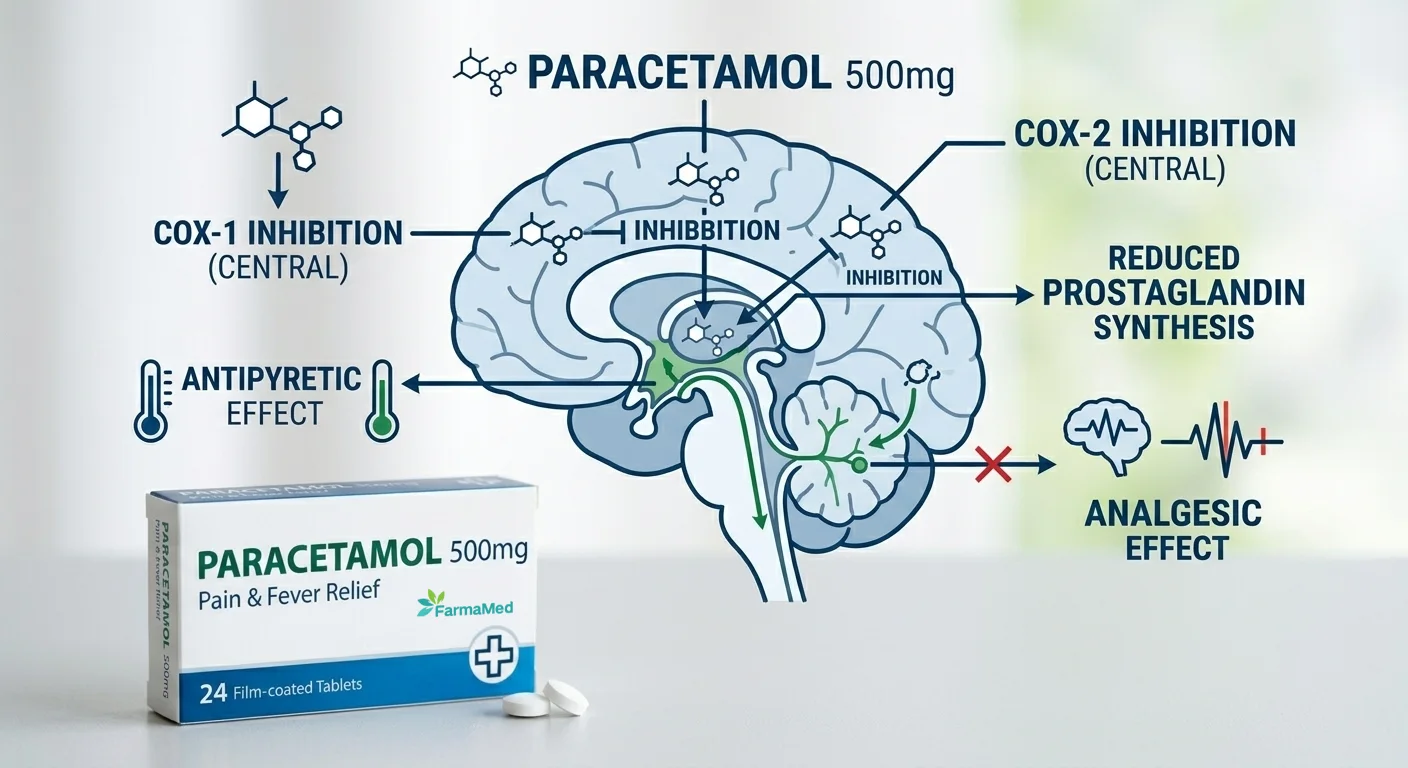
oral tablets manufacturer
An oral tablets manufacturer represents a specialized pharmaceutical production facility dedicated to creating solid dosage forms for oral administration. These sophisticated manufacturing operations combine advanced technology, rigorous quality control systems, and comprehensive regulatory compliance to produce tablets that meet stringent pharmaceutical standards. The primary function of an oral tablets manufacturer centers on transforming raw pharmaceutical ingredients into precisely formulated, consistently dosed tablets through carefully controlled processes. Modern oral tablets manufacturer facilities utilize state-of-the-art equipment including high-speed tablet presses, granulation systems, coating machines, and automated packaging lines to ensure optimal production efficiency and product quality. The technological features of contemporary oral tablets manufacturer operations include real-time monitoring systems, computerized process controls, and sophisticated analytical testing equipment that validates every batch produced. These manufacturers employ various tablet formation techniques such as direct compression, wet granulation, and dry granulation, selecting the most appropriate method based on the specific characteristics of the active pharmaceutical ingredient and desired tablet properties. Quality assurance protocols within oral tablets manufacturer facilities encompass comprehensive testing for content uniformity, dissolution rates, hardness, friability, and disintegration times. The applications of oral tablets manufacturer services extend across multiple therapeutic areas including cardiovascular medications, antibiotics, pain management drugs, vitamins, dietary supplements, and specialty pharmaceuticals. Modern oral tablets manufacturer facilities also provide custom formulation development, regulatory support, and packaging solutions tailored to specific client requirements. These manufacturers serve diverse markets including prescription pharmaceuticals, over-the-counter medications, nutraceuticals, and generic drug markets. The oral tablets manufacturer industry continues evolving with innovations in continuous manufacturing processes, advanced coating technologies, and personalized medicine approaches that enable more efficient production of targeted therapeutic solutions for global healthcare needs.