Customizable Formulation Capabilities in Magnesium Citrate Tablets Production
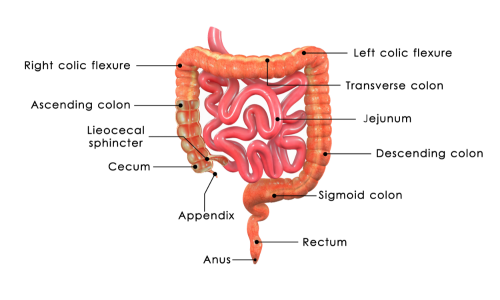
The customizable formulation capabilities inherent in modern magnesium citrate tablets production enable manufacturers to create targeted supplements that address specific health needs, dietary restrictions, and demographic requirements. This flexibility begins with the ability to adjust elemental magnesium content across a wide range, typically from 100mg to 400mg per tablet, allowing healthcare providers to prescribe precise dosages that align with individual patient requirements and therapeutic goals. The magnesium citrate tablets production process accommodates various tablet sizes and shapes, from small, easy-to-swallow tablets for elderly patients to larger, scored tablets that can be divided for flexible dosing options. Specialized coating technologies employed in magnesium citrate tablets production can create enteric-coated formulations that resist stomach acid and dissolve in the intestinal tract, reducing gastrointestinal irritation while maintaining optimal absorption rates. The production capabilities extend to creating combination formulations that incorporate complementary nutrients such as vitamin D3, calcium, or B-vitamins, providing comprehensive mineral support in a single tablet format. For consumers with specific dietary needs, the magnesium citrate tablets production process can accommodate vegetarian and vegan formulations using plant-based excipients and coatings, while avoiding common allergens like gluten, dairy, and soy. Extended-release formulations represent another significant customization option, where the magnesium citrate tablets production process incorporates specialized matrix systems that control magnesium release over 8-12 hours, providing sustained therapeutic levels while minimizing peak-related side effects. The flavor-masking capabilities integrated into magnesium citrate tablets production address the naturally bitter taste of magnesium compounds, utilizing coating technologies and flavoring systems that improve palatability without compromising bioavailability. Color customization options allow manufacturers to create distinctive product identification through the magnesium citrate tablets production process, while maintaining product integrity and stability. These formulation capabilities also extend to packaging innovations, including blister packs for improved stability, bottle packaging for bulk quantities, and unit-dose packaging for clinical applications. The versatility of magnesium citrate tablets production enables manufacturers to respond quickly to market demands, regulatory changes, and emerging scientific research, ensuring that consumers have access to the most current and effective magnesium supplementation options available in the marketplace.