Professional-Grade Quality and Safety Standards
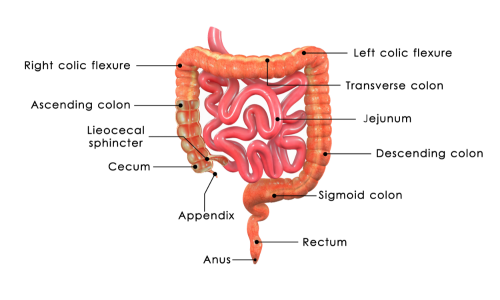
Pharmaceutical magnesium citrate tablets are manufactured according to the highest industry standards, ensuring exceptional quality, purity, and safety that healthcare professionals and consumers can trust with confidence. The production process begins with pharmaceutical-grade raw materials that undergo rigorous testing for identity, purity, and potency before being approved for use in manufacturing. Each batch of pharmaceutical magnesium citrate tablets is produced in facilities that comply with Good Manufacturing Practices regulations, which mandate strict protocols for cleanliness, equipment maintenance, and quality control procedures. Advanced analytical testing methods, including high-performance liquid chromatography and atomic absorption spectroscopy, verify the exact magnesium content and confirm the absence of harmful contaminants such as heavy metals, microorganisms, and residual solvents. The tablet compression process utilizes precision equipment that ensures uniform weight, thickness, and hardness across all units, guaranteeing consistent dosing and dissolution characteristics. Quality assurance protocols include stability testing under various environmental conditions to confirm that pharmaceutical magnesium citrate tablets maintain their potency and safety throughout their stated shelf life. Third-party laboratory verification provides additional assurance of quality and compliance with label claims, giving consumers confidence in the accuracy of product specifications. The packaging systems protect pharmaceutical magnesium citrate tablets from moisture, light, and oxygen exposure that could degrade the active ingredients over time. Traceability systems allow for complete tracking of each batch from raw material sourcing through final product distribution, enabling rapid response if any quality issues arise. Safety testing includes evaluation of potential allergens, ensuring that pharmaceutical magnesium citrate tablets are suitable for individuals with common food sensitivities. The manufacturing facilities undergo regular inspections by regulatory authorities to maintain their certifications and ensure ongoing compliance with evolving safety standards. Documentation and record-keeping procedures create a comprehensive quality history for each production batch, supporting transparency and accountability in the manufacturing process. This commitment to professional-grade quality standards ensures that pharmaceutical magnesium citrate tablets deliver consistent, reliable results while maintaining the highest levels of safety for long-term use.