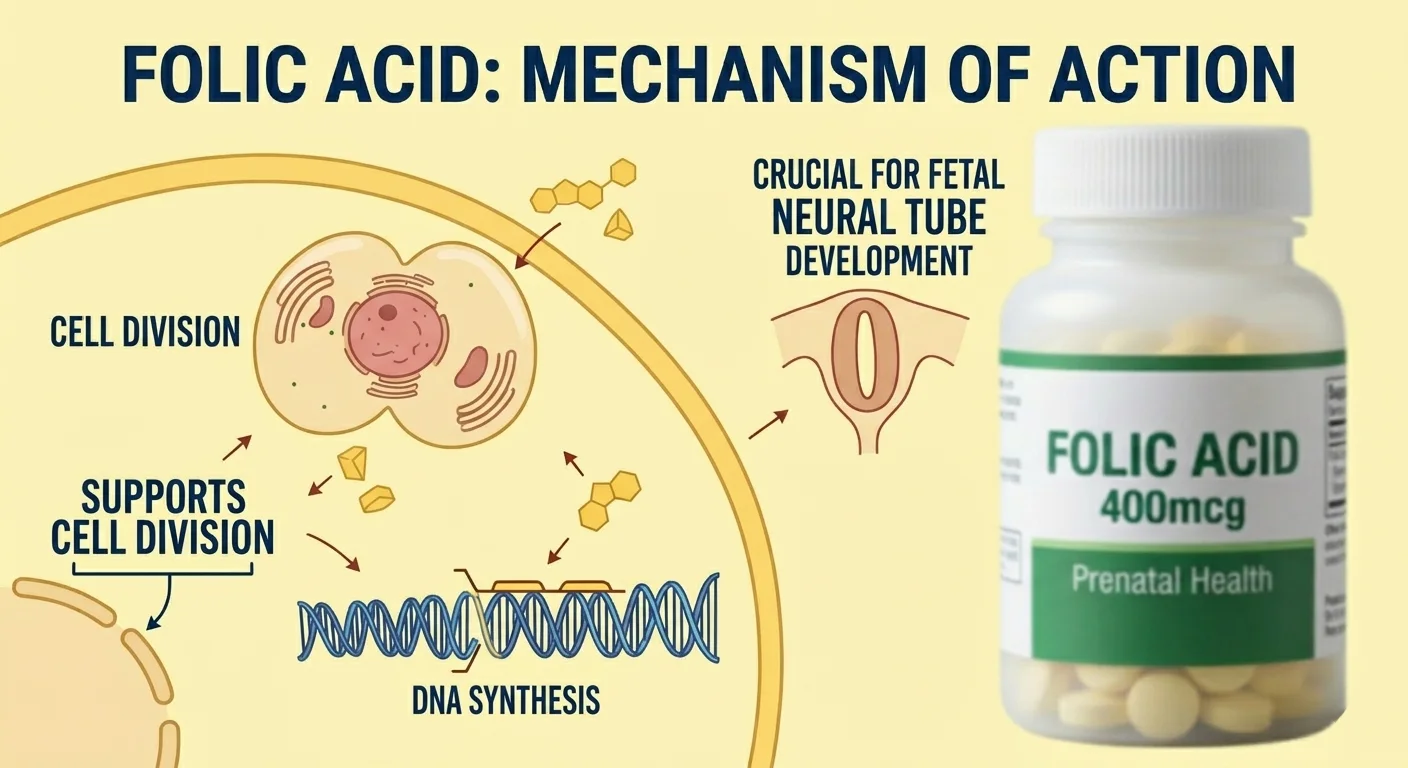
Comprehensive Regulatory Expertise and Global Compliance
Navigating the complex landscape of pharmaceutical regulations requires specialized knowledge and experience that professional ferrous folic acid tablets supplier companies have developed through years of international operations and regulatory interactions. This expertise represents a crucial advantage for businesses seeking to market nutritional supplements across multiple jurisdictions with varying regulatory frameworks. Leading suppliers maintain dedicated regulatory affairs teams staffed with experts who understand the nuances of pharmaceutical regulations in major markets including the United States, European Union, Canada, Australia, and emerging markets throughout Asia and Latin America. These professionals stay current with evolving regulatory requirements, ensuring that product formulations, labeling, and manufacturing processes remain compliant with the latest standards. The regulatory expertise of established ferrous folic acid tablets supplier companies extends beyond mere compliance to encompass strategic guidance that helps customers optimize their market entry strategies and product positioning. This includes assistance with product registration processes, clinical documentation requirements, and labeling optimization to meet local market preferences while maintaining regulatory compliance. Professional suppliers maintain comprehensive regulatory documentation packages that include stability studies, bioavailability data, and safety assessments that support product approvals in new markets. This documentation represents significant value for customers, as developing these materials independently would require substantial time and financial investment. Quality system certifications such as ISO certification, cGMP compliance, and facility inspections by international regulatory authorities provide additional assurance of supplier credibility and product quality. These certifications undergo regular audits and renewals, ensuring ongoing compliance with evolving standards. The regulatory expertise also encompasses understanding of specific requirements for different market segments, such as dietary supplements, prescription medications, and over-the-counter products, enabling suppliers to provide appropriate guidance for optimal product classification and marketing strategies. This comprehensive regulatory support reduces market entry barriers, accelerates time-to-market, and minimizes compliance risks for customers expanding into new geographic markets or product categories.