hydrocortisone cream production
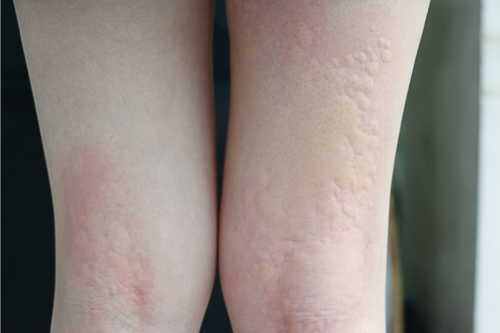
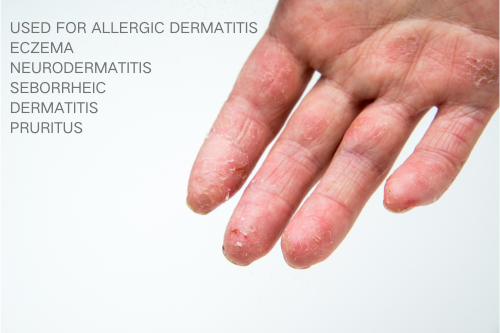
Hydrocortisone cream production represents a sophisticated pharmaceutical manufacturing process that delivers essential topical corticosteroid medications to patients worldwide. This complex production system involves precise formulation, sterile manufacturing environments, and stringent quality control measures to ensure therapeutic efficacy and patient safety. The main functions of hydrocortisone cream production encompass creating anti-inflammatory topical treatments that effectively reduce skin irritation, redness, and swelling associated with various dermatological conditions including eczema, dermatitis, and allergic reactions. The technological features of modern hydrocortisone cream production facilities incorporate advanced mixing systems, homogenization equipment, and automated filling machines that maintain consistent product quality throughout the manufacturing cycle. State-of-the-art production lines utilize computerized process controls, temperature monitoring systems, and contamination prevention protocols to ensure every batch meets pharmaceutical standards. The applications of hydrocortisone cream production extend across multiple therapeutic areas, serving patients who require reliable anti-inflammatory treatments for chronic skin conditions, acute allergic reactions, and post-procedural care. Manufacturing facilities employ specialized emulsification technology that creates stable cream formulations with optimal skin penetration properties, ensuring effective delivery of active pharmaceutical ingredients. Quality assurance protocols integrated into hydrocortisone cream production include microbiological testing, potency verification, and stability studies that guarantee product shelf life and therapeutic performance. The production process incorporates advanced packaging systems that preserve product integrity while providing convenient application methods for end users. Environmental controls within manufacturing facilities maintain appropriate humidity, temperature, and air quality standards essential for pharmaceutical-grade production.